INTRODUCTION
Antibody-mediated rejection (ABMR) is a major cause of graft dysfunction and loss in renal transplantation. It is characterized by donor-specific antibodies (DSAs), complement activation, and microvascular injury. Despite advances in immunosuppression, ABMR remains difficult to treat and is associated with poor long-term outcomes.
CASE PRESENTATION
Mr. V a 24-year-old male with chronic kidney disease secondary to chronic interstitial nephritis, underwent swap renal transplantation on 19/05/2025. Pre-transplant workup revealed negative virtual crossmatch and flow cytometry crossmatch, but donor-specific antibodies were positive (Class I B35, Class II DP and DQ). Post-transplant renal function was initially stable.
Early graft dysfunction was suspected to be acute cellular rejection; however, a biopsy on 03/06/2025 revealed acute tubular necrosis (ATN) secondary to calcineurin inhibitor toxicity. Despite dose adjustments and addition of sirolimus, serum creatinine rose to 1.83 mg/dL, prompting repeat biopsy.
HISTOPATHOLOGY FINDINGS
Renal allograft biopsy (06/03/2026) demonstrated:
- Peritubular capillaritis (ptc3) without glomerulitis or transplant glomerulopathy.
- Moderate interstitial fibrosis and tubular atrophy (25–30%) with lymphoid aggregates.
- Mild acute tubular injury.
- C4d focal positivity (C4d1, <10% peritubular capillaries).
- Direct immunofluorescence negative for IgG, IgA, IgM, C3c, C1q.
IMPRESSION: Features suggestive of active ABMR, with recommendation for correlation with circulating DSAs.
DISCUSSION
According to the Banff 2019 criteria, ABMR diagnosis requires:
- Histologic evidence of microvascular injury (glomerulitis, peritubular capillaritis).
- Serologic evidence of DSAs.
- Immunopathologic evidence (C4d positivity).
This patient demonstrated peritubular capillaritis and focal C4d positivity, with known DSAs, fulfilling criteria for ABMR. The presence of moderate IFTA indicates chronic injury, which may limit reversibility.
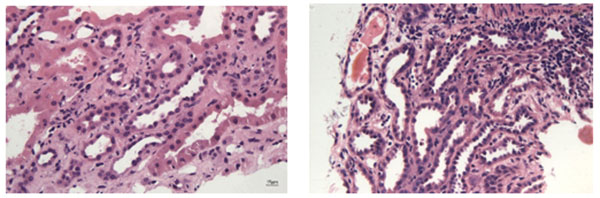
Figure 1 – Histopathological slide of Mr V, with ABMR changes- peritubular capillaritis and chronic changes
PATHOPHYSIOLOGY
ABMR results from DSAs binding to donor HLA antigens, activating complement, and causing endothelial injury. Chronic ABMR leads to transplant glomerulopathy and progressive graft loss.
MANAGEMENT
- Acute ABMR: Plasmapheresis, IVIG, rituximab, bortezomib, and complement inhibitors (eculizumab).
- Chronic ABMR: Optimization of immunosuppression, IL-6 blockade (tocilizumab), and experimental therapies such as imlifidase.
- In this case, close monitoring of DSAs and renal function, with consideration of antibody-targeted therapy, is warranted.
CONCLUSION
This case highlights the diagnostic complexity of post-transplant dysfunction, where overlapping etiologies such as calcineurin inhibitor toxicity and ABMR coexist. Vigilant monitoring, timely biopsy, and correlation with DSA levels are essential for guiding therapy and improving graft survival.
REFERENCES
- Loupy A, Lefaucheur C. Antibody-mediated rejection of solid-organ allografts. N Engl J Med. 2018;379(12):1150–1160.
- Haas M, Loupy A, Lefaucheur C, Roufosse C, Glotz D, Seron D, et al. The Banff 2019 Kidney Meeting Report: Revised diagnostic criteria for chronic active antibody-mediated rejection. Kidney Int. 2020;97(6):1113–1126.
- Stegall MD, Diwan T, Raghavaiah S, Cornell LD, Burns JM, Dean PG, et al. Terminal complement inhibition decreases antibody-mediated rejection in sensitized renal transplant recipients. Am J Transplant. 2011;11(11):2405–2413.
- Jordan SC, Lorant T, Choi J, Kjellman C, Winstedt L, Bengtsson M, et al. IgG endopeptidase in highly sensitized patients undergoing transplantation. N Engl J Med. 2017;377(5):442–453.
Colvin RB, Smith RN. Antibody-mediated organ-allograft rejection. Nat Rev Immunol. 2005;5(10):807–817.

Dr. C. Chaitra
DrNB Resident
Kauvery Hospital, Alwarpet Chennai.[1]