Abstract
Trauma-related hemorrhage is a leading cause of preventable mortality. While torso bleeding is commonly prioritized, extremity hemorrhage can be equally life-threatening and often under-recognized.
we present a 59-year-old male presented following a road traffic accident with bilateral lower limb injuries. Initial assessment revealed stable vitals with no active external bleeding. Despite negative EFAST and imaging for thoracoabdominal injuries, the patient developed sudden hypotension. Serial reassessment revealed occult hemorrhage from a left lower limb vascular injury involving the posterior tibial artery.
Conclusion: This case highlights the importance of serial monitoring, early recognition of occult extremity bleeding, and timely activation of massive transfusion protocols in trauma care.
Keywords
Extremity hemorrhage, trauma, vascular injury, hypovolemic shock, massive transfusion
Introduction
Hemorrhage is the leading cause of preventable mortality in trauma. While torso bleeding is commonly emphasized, extremity hemorrhage can be significant and occult, leading to delayed recognition and deterioration.
Case Presentation
A 59-year-old male presented to the emergency department with an alleged history of road traffic accident (two-wheeler vs two-wheeler). He sustained injuries to head , both lower limbs and had a history of loss of consciousness and amnesia of the event. He was initially taken to an outside hospital, and details of prior management were not available.
There was no history of chest pain, breathlessness, abdominal pain, or bowel and bladder disturbances.
Past medical history : hepatitis B positive , Rheumatoid arthritis
Past surgical history: S/P liver transplantation (2022)
Primary Survey (XABCDE)
X (Exsanguination): No active external bleeding noted. Left leg dressing was intact with no soakage.
A (Airway): Airway patent. Cervical spine immobilization applied.
B (Breathing): Bilateral air entry present, SpO₂: 98% on room air, RR: 18/min
C (Circulation): S1, S2 present, no murmur, BP: 100/60 mmHg, PR: 76 bpm
Abdomen: Soft, non-tender, bowel sounds present
D (Disability): GCS: 15/15, Pupils: Equal and reactive (2 mm), Moving all four limbs
E (Exposure): Temperature: 97.6°F
Adjuncts
ABG
PH: 7.36
PCO₂: 32 mmHg
PO₂: 88 mmHg
HCO₃⁻: 18.3 mmol/L
Base excess: -7.3
Lactate: 4.4 mmol/L
Hemoglobin: 9.5 g/dL
Hematocrit: 24%
Sodium: 132 mEq/L
Potassium: 3.5 mEq/L
Chloride: 105 mEq/L
Creatinine: 1.4 mg/dL
ECG: Normal sinus rhythm, no acute ST-T changes
EFAST:
B/L lung sliding present
No pneumothorax or hemothorax
Cardiac contractility : adequate no cardiac tamponade
No free fluid in hepatorenal and splenorenal angle
Bladder normal.
Secondary Survey
head-to-toe examination revealed:
Head & Face: Forehead: abrasion of 3x3cm above the right eyebrow.
Neck: No tenderness or swelling
Chest: Bilateral air entry equal, no tenderness
Abdomen: Soft, non-tender, no distension
Pelvis: Stable
Spine: No deformity or tenderness
Extremities
Left Lower Limb:
Deep laceration (~20 × 5 × 10 cm) over anterolateral aspect of lower leg
Muscle exposure noted
No active bleeding
Restricted ankle movement
Painful knee movement
Distal pulses +
Right Lower Limb:
Knee swelling +
Tenderness+
ROM Restricted and painful
Crepitus present
Distal pulses palpable
No additional injuries were identified.
Initial management in ED
- Inj Tetanus toxoid 0.5 ml IM stat
- IV fluids: 250 ml bolus followed by 100ml/hr
- Broad-spectrum antibiotics
- Wound irrigation with 1L saline
- Sterile compression dressing applied
- B/L lower limbs immobilized with splint
- Orthopedic and plastic surgery consultation obtained
- CT brain with C spine
XRAY for B/L lower limb ordered
Clinical Course
Upon awaiting for radiological imaging Patient developed hypotension
On reassessment:
Blood pressure dropped to 70/40 mmHg
Repeat EFAST: No new findings
IVC: Collapsible
Intervention
Fluid resuscitation initiated
Massive transfusion protocol (1:1:1) activated
Noradrenaline infusion started
Patient shifted for PAN-CT – TRAUMA PROTOCOL
- CT Brain: Tiny hemorrhagic contusion in left inferior temporal cortex
Minimal subarachnoid hemorrhage - Chest & Abdomen: No hemothorax, pneumothorax, or hemoperitoneum
Incidental cholelithiasis - Spine: No fractures
X-rays:
Right knee: Proximal tibia fracture
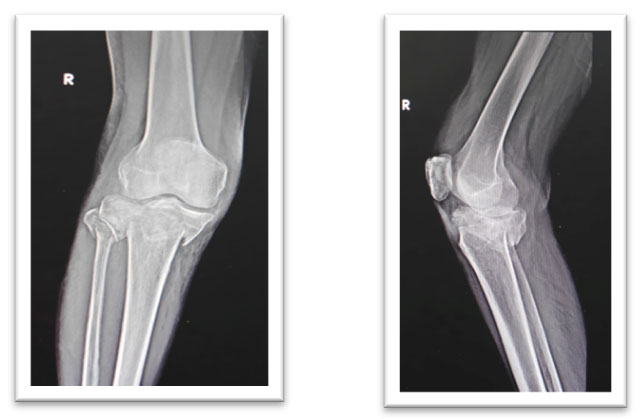
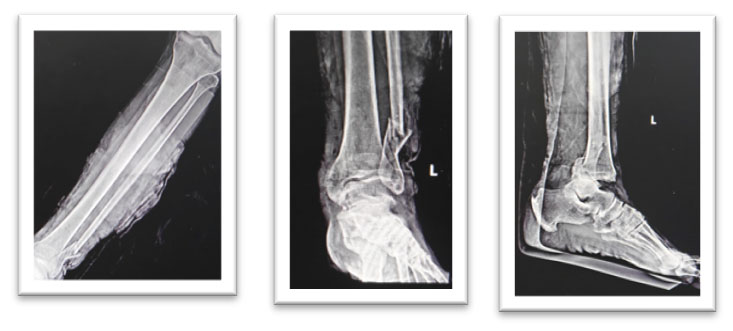
Left ankle: Compound trimaleolar ankle fracture
Clinical Deterioration
Identification of Bleeding Source : With no torso source identified: upon exploration of left leg
Active oozing observed
Wound packed and compressed
New finding: Distal pulse became absent
An urgent vascular opinion obtained
At this point BP :90/60 mm hg/ PR: 90 bpm, on ongoing PRBC transfusion.
- CT Peripheral Angiography:
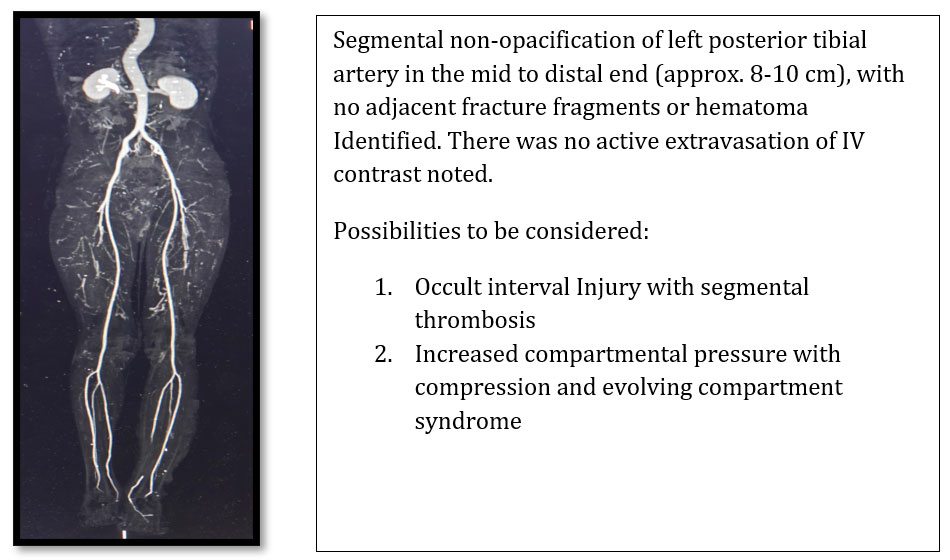
IMPRESSION: Compound trimaleolar fracture left ankle with Vascular injury
DIFFINITIVE MANAGEMENT: Patient shifted to OT with ongoing resuscitation.
- Vascular: Left posterior tibial artery repair done .
- Plastic Surgery: wound debridement +SSG done
- The patient was stabilized and underwent staged orthopedic management, including left ankle spanning exfix on day 1, followed by right proximal dual column tibial plating on day 5
OUTCOME:
Postoperatively, the patient remained hemodynamically stable with satisfactory clinical recovery and was discharged in a stable condition on day 7.
Discussion
This case highlights a critical and often under-recognized cause of traumatic shock—occult extremity hemorrhage leading to delayed exsanguination despite an initially stable presentation. While trauma protocols traditionally emphasize thoracic, abdominal, and pelvic sources of bleeding, this case reinforces that extremities can serve as a significant and potentially fatal reservoir of blood loss, particularly in the presence of deep soft tissue and vascular injury.
A key teaching point illustrated here is the fundamental trauma principle that “in trauma, shock must be presumed hemorrhagic until proven otherwise.” In this patient, the absence of active external bleeding, combined with a negative EFAST and unremarkable torso imaging, initially suggested hemodynamic stability. However, the subsequent development of hypotension with elevated lactate and base deficit indicated ongoing occult hypoperfusion. This underscores that early clinical findings may be deceptively reassuring, and reliance on a single-point assessment can delay recognition of life-threatening hemorrhage.
The pathophysiology of delayed bleeding in this case is multifactorial. Initial hemostasis may have been achieved through temporary tissue tamponade and unstable clot formation within the deep muscular compartments of the leg. Subsequent resuscitation, along with patient handling and interdepartmental transfers, may have contributed to clot dislodgement, converting a contained hematoma into active hemorrhage. This phenomenon is well described in trauma but remains underappreciated in routine clinical practice. Therefore, any clinical deterioration following patient movement should prompt immediate reassessment of previously identified injuries.
Another important aspect is the dynamic nature of vascular injuries. The initial presence of distal pulses in this patient likely reflected collateral circulation or partial arterial injury, which masked the severity of vascular compromise. The later loss of distal pulses was a crucial clinical sign that prompted further evaluation. This highlights that the presence of distal pulses does not exclude significant vascular injury, and serial neurovascular examinations are essential in extremity trauma.
This case also emphasizes the limitations of imaging modalities in early trauma assessment. Both EFAST and PAN-CT failed to identify a bleeding source. Notably, the PAN-CT protocol in our setting included imaging only up to the mid-thigh and did not cover distal extremities, thereby potentially missing vascular injuries below this level. Additionally, the hemorrhage was localized and confined within soft tissue compartments without significant accumulation detectable on imaging. This highlights that negative imaging does not exclude ongoing hemorrhage, particularly in extremity trauma, and reinforces that clinical judgment and repeated bedside reassessment remain paramount in identifying evolving bleeding sources.
The timely activation of the massive transfusion protocol (MTP) played a pivotal role in this patient’s survival. Early balanced transfusion (1:1:1 ratio) helps mitigate trauma-induced coagulopathy, restore circulating volume, and improve oxygen delivery. Importantly, this case reinforces that MTP should be initiated based on clinical suspicion rather than delayed until definitive identification of the bleeding source.
From a systems perspective, this case underscores the importance of a multidisciplinary approach, involving emergency physicians, orthopedic surgeons, vascular surgeons, and plastic surgeons. Early coordination allowed for definitive surgical control of bleeding, including vascular ligation and fracture stabilization, which ultimately resulted in a favorable outcome.
Several key learning points emerge from this case:
- Extremities are a major but often overlooked source of hemorrhage in trauma
- Absence of active external bleeding does not exclude significant blood loss
- Serial reassessment is critical and often lifesaving
- Patient movement and transfers can precipitate delayed hemorrhage
- Distal pulses may be present despite significant vascular injury
- Negative EFAST and imaging do not rule out ongoing bleeding
- Early activation of MTP improves outcomes in suspected hemorrhagic shock
Conclusion
This case reinforces that occult extremity hemorrhage is a potentially fatal yet preventable cause of delayed shock in trauma patients. Emergency physicians must maintain a high index of suspicion, particularly in patients with unexplained hypotension and negative torso imaging. Ultimately, vigilant serial reassessment, early resuscitation, and prompt surgical intervention remain the cornerstones of successful trauma management.
References
- American College of Surgeons. Advanced Trauma Life Support (ATLS®): Student Course Manual. 11th Chicago, IL: American College of Surgeons; 2025.
- Kauvar DS, Lefering R, Wade CE. Impact of hemorrhage on trauma outcome: an overview of epidemiology, clinical presentations, and therapeutic considerations. Ann Surg. 2006;243(5):622–631.
- Eastridge BJ, Mabry RL, Seguin P, et al. Death on the battlefield (2001–2011): implications for the future of combat casualty care. J Trauma Acute Care Surg. 2012;73(6 Suppl 5):S431–S437.
- Fox N, Rajani RR, Bokhari F, et al. Evaluation and management of lower extremity arterial trauma: An Eastern Association for the Surgery of Trauma (EAST) practice management guideline. J Trauma Acute Care Surg. 2012;73(5 Suppl 4):S315–S320.
- Feliciano DV. Management of peripheral arterial injury. Curr Opin Crit Care. 2010;16(6):602–608.
- Tisherman SA, Barie PS, Bokhari F, et al. Clinical practice guideline: endpoints of resuscitation. J Trauma. 2004;57(4):898–912.

Dr. Avinash S
Department of Emergency Medicine,
Kauvery Hospital, Alwarpet, Chennai.[1]

Dr . Ashok Nandagopal
HOD, Department of Emergency Medicine,
Kauvery Hospital, Alwarpet, Chennai.[1]