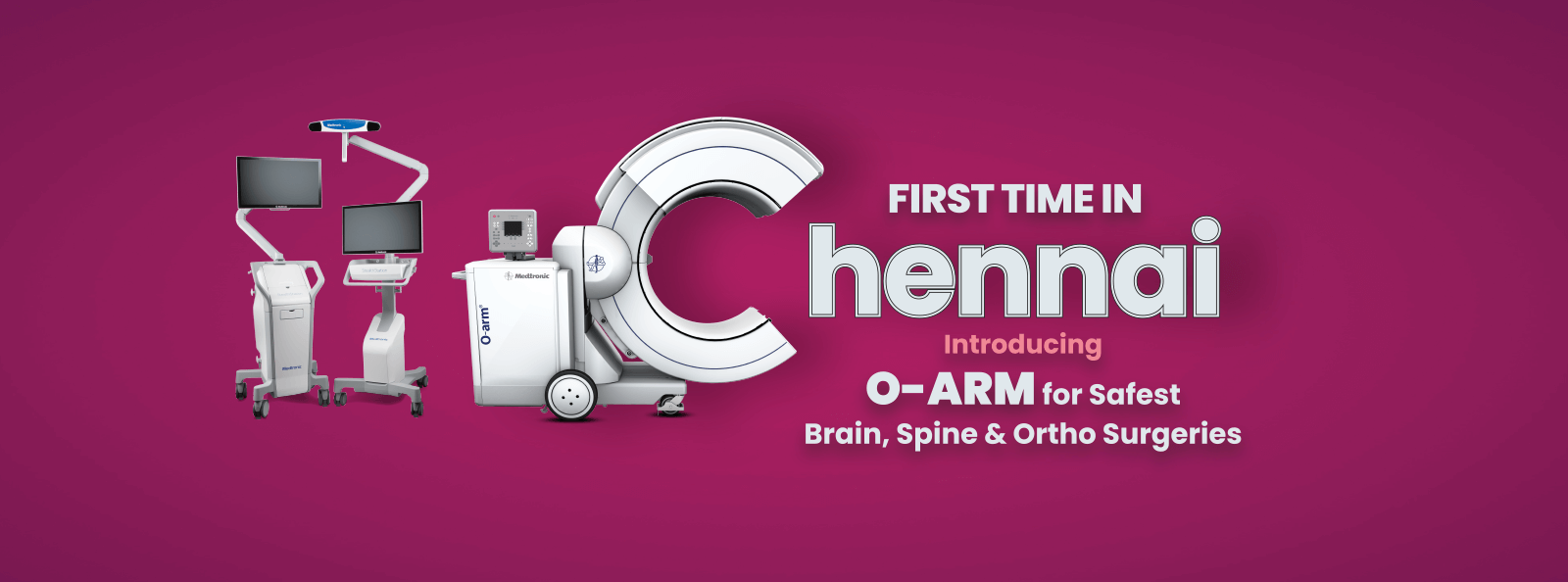
Why Kauvery Hospital…
We are a leading, multi-speciality hospital offering best-in-class medical services for nearly 2 decades. With paramount focus on patient care, patient safety and patient satisfaction, we are committed to offer exemplary medical services going beyond the norm of healthcare.

Empathy driven personalized patient care.

Socially conscious organization

Vastly experienced team of doctors with extensive clinical expertise

Dedicated and exclusive patient advice and liaison team

High quality medical services made affordable

Dedicated nursing and paramedical support
For International patients
At Kauvery Hospital our services are designed to provide a very special environment to enhance healing and encompassing the comfort of your home away from home. Our team comprising of International Concierges, Interpreters, Billing Assistance and other associated non-medical staff are all trained to provide assistance efficiently.