CAR- T cell therapy
Background
- In 1980s, the concept of using T cells to fight against cancer emerged.
- In the 1990s the development of Chimeric Antigen Receptor (CAR)- T cell therapy came as a crucial breakthrough in treatment of cancers.
- CAR- T cell therapy helps the immune system to recognize and kill cancer cells.
- Chimeric means that two or more different genes, from mouse and human sources, are combined to create a new mixed gene which can recognize a particular target antigen.
- It is in use for the treatment of multiple myeloma and certain types of Leukemia and Lymphoma.
Images: BMS (Bristol Myers Squibb)
Discussion
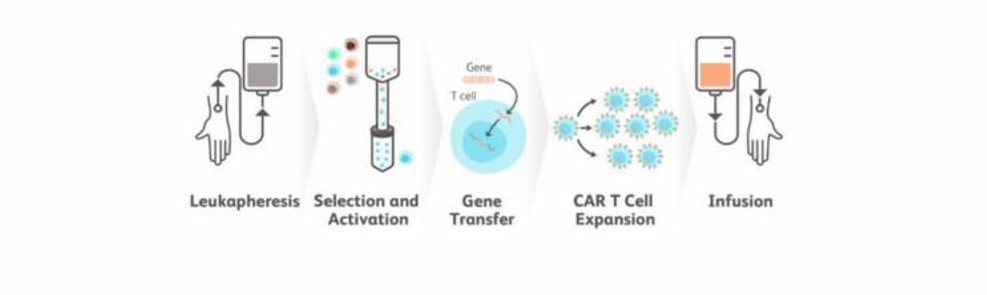
- CAR- T cell therapy begins by removing white blood cells from blood through a process known as apheresis.
- The removed T cells are modified genetically by adding a protein- designed gene CAR.
- The modified CAR -T cells are infused back into the bloodstream of the patient where they target cancer cells and destroy them.
- CAR- T cell therapy also has certain limitations, due to its adverse effects like neurotoxicity and cytokine release syndrome (CRS) etc.
- In 2023, India’s first CAR- T cell therapy called NexCAR19 received its approval for treating B cell lymphomas and B Cell Acute Lymphoblastic Leukemia.