Hypoglycemic encephalopathy with diffuse cerebral edema and multi‑organ dysfunction: A clinical pharmacist perspective on a patient with type 2 diabetes mellitus
Mohamed Asir*, Berlin
Department of Clinical Pharmacy, Kauvery Hospital, Tirunelveli, Tamil Nadu
*Correspondence
Abstract
Hypoglycemic encephalopathy is a rare but catastrophic neurological complication of severe or prolonged hypoglycemia, often associated with poor prognosis, particularly in patients with underlying endocrine comorbidities. We report the case of a 59‑year‑old male with Type 2 Diabetes Mellitus and longstanding hypothyroidism who presented with sudden unresponsiveness following recurrent episodes of slurred speech over six months. On initial evaluation, he was found to have profound hypoglycemia (random blood glucose 39 mg/dL) and a Glasgow Coma Scale score of 3/15. Despite prompt glucose correction and aggressive intensive care management, the patient remained comatose. Neuroimaging demonstrated diffuse cerebral edema with features suggestive of hypoxic–ischemic encephalopathy. His clinical course was further complicated by systemic inflammatory response, acute kidney injury, hyperammonaemia, metabolic acidosis, anemia, and cardiovascular instability, culminating in multiorgan dysfunction. The patient failed to demonstrate neurological recovery and succumbed to his illness despite prolonged ventilatory and supportive management. This case highlight’s role of hypothyroidism may be an under‑recognised factor for recurrent and prolonged hypoglycemia, leading to severe neuroglycopenic injury. Early recognition of subtle hypoglycemic symptoms and optimisation of endocrine comorbidities are essential to prevent irreversible neurological damage and fatal outcomes in high‑risk diabetic patients.
Keywords: Hypoglycemic encephalopathy, cerebral edema; Hypothyroidism; Type 2 diabetes mellitus; Multiorgan dysfunction; Hypoxic–ischemic brain injury
Introduction
Hypoglycemia is a serious and potentially life‑threatening complication of diabetes mellitus, particularly in elderly patients with long‑standing disease and associated endocrine comorbidities.[1] Severe or prolonged hypoglycemia can result in neuroglycopenia, leading to altered sensorium, seizures, cerebral edema, hypoxic–ischemic brain injury, and irreversible neurological damage. Hypoglycemic encephalopathy is characterised by persistent impairment of consciousness despite correction of blood glucose levels and is often associated with poor neurological outcomes.[2]
Hypothyroidism is often under‑recognised contributor to hypoglycemia.[3] Thyroid hormone deficiency impairs hepatic gluconeogenesis and glycogenolysis, reduces insulin clearance, and reduces counter‑regulatory hormonal responses, predisposing patients to recurrent and prolonged hypoglycemia.[4] Attenuation of autonomic warning symptoms may delay recognition, allowing progression to severe neuroglycopenia, in which manifestations such as slurred speech and altered mental status may reflect metabolic encephalopathy rather than structural brain disease.[5]
When severe hypoglycemia occurs in the setting of hypothyroidism, the risk of diffuse cerebral edema and hypoxic–ischemic injury is significantly increased, especially in patients with additional systemic insults such as infection, renal dysfunction, or cardiovascular instability.[6] This case report describes a fatal instance of hypoglycemic encephalopathy in a patient with type 2 diabetes mellitus and longstanding hypothyroidism, highlighting the potential role of thyroid dysfunction as a predisposing factor and underscoring the importance of early recognition and preventive endocrine evaluation to avert catastrophic neurological outcomes.
Case Presentation
A 59‑year‑old male, known case of type 2 diabetes mellitus and hypothyroidism, with a past history of left foot debridement done in 2023, history of pyelonephritis and slurred speech on and off for past six months was brought to the emergency department after being found unresponsive at home at around 9:00 PM on 17.02.2026. The patient was last seen conscious on 16.02.2026, when he was noted to have slurred speech. He was initially taken to a nearby hospital, where he was found to have a Glasgow Coma Scale (GCS) score of 3/15 and severe hypoglycemia with a random blood glucose level of 39 mg/dL. He received intravenous 25% dextrose and was referred to our hospital for further management. On arrival at our centre, the patient remained unconscious and afebrile, with a random blood glucose level of 79 mg/dL. In view of persistent poor sensorium and low GCS, the patient was endotracheally intubated and connected to mechanical ventilation.
Past Medical History
The patient had a known history of type 2 diabetes mellitus and hypothyroidism, with a past history of left foot debridement performed in 2023 and pyelonephritis, along with intermittent slurred speech over the preceding six months; there was no previously documented history of cerebrovascular accident, seizure disorder, or chronic kidney disease.
Clinical Course and Management
Hypoglycemia was managed with intravenous 25% dextrose and insulin infusion, with blood sugar monitored regularly and diabetes managed according to blood sugar levels. Routine laboratory investigations revealed elevated total leukocyte count, increased C‑reactive protein, and elevated procalcitonin levels, suggestive of systemic infection or inflammatory response. Neurological evaluation was undertaken, and neurologist opinion was obtained. Electroencephalography (EEG) showed intermittent polyspike, wave discharge noted and posterior predominant alpha rhythm. CT brain, and MRI brain were performed, which demonstrated diffuse cerebral edema. Neurologist’s advice was followed. The patient was treated with intravenous antibiotics, anticonvulsants, intravenous fluids (5% dextrose), anticoagulants, antiplatelets, osmotic diuretics, anti‑inflammatory drugs, vitamin supplements, proton pump inhibitors, nebulisation, and other supportive care measures.
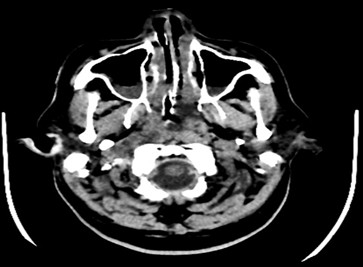
Fig (1): CT brain plain
On 20.02.2026, the patient developed hypertension and started on antihypertensive medications. Renal parameters worsened with serum creatinine rising to 2.50 mg/dL. Nephrologist opinion was obtained, and treatment was initiated in view of unexplained renal dysfunction. Cardiologist opinion was also obtained, and echocardiography was performed. Ophthalmology consultation was done. Repeat total leukocyte count on 20.02.2026 showed further increase, and intravenous antibiotics were escalated to Inj. Meropenem 500 mg three times daily. The patient developed hypotension with decreased systolic arterial pressure, and a left internal jugular vein line was secured.
Intensive care course and complications
On 23.02.2026, the patient developed tachypnea, tachycardia, and hypotension, for which vasopressin infusion was initiated. Antihypertensive medications were withheld on the same day due to hypotension. Urine culture, endotracheal culture, and blood culture showed no growth. The patient remained on mechanical ventilation, and intravenous antibiotics were cont
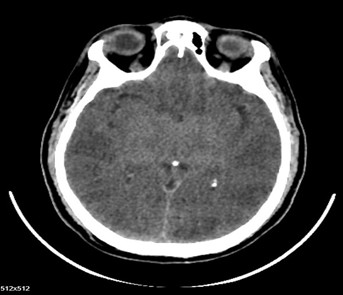
inued. Vasopressin gradually weaned off based on blood pressure. Serum ammonia levels were found to be elevated (140 µmol/L) and were managed with syrup Duphalac. A repeat CT brain performed on 24.02.2026 showed probable sequelae of hypoxic‑ischemic insult. In view of cerebral findings, anticoagulants were stopped and antiplatelets were withheld from 24.02.2026. Trial weaning from mechanical ventilation was attempted but was unsuccessful. The patient continued to require ventilatory support. Poor neurological prognosis was repeatedly explained to the attenders.
Fig (2): CT brain showed probable sequelae of hypoxic‑ischemic insult
Subsequent course in the hospital
On 27.02.2026, renal parameters, electrolytes, and ammonia levels improved. The patient remained on mechanical ventilation and was planned for tracheostomy, which was performed on 01.03.2026. Repeat serum albumin showed hypoalbuminemia (2.52 g/dL), and intravenous albumin transfusion was given. On 02.03.2026, hemoglobin dropped to 6.8 g/dL, and one unit of packed red blood cells was transfused. Repeat PRBC transfusion was administered subsequently. Planned weaning from mechanical ventilation was attempted with intermittent BIPAP support.
Renal parameters showed improvement. Neurological monitoring continued. The patient remained on BIPAP support. Arterial blood gas analysis showed borderline metabolic acidosis. In view of malena, medical gastroenterology opinion was obtained, and Inj. Pantocid infusion was advised. Neurologist opinion was again obtained, and poor prognosis was reiterated to the attenders. The patient was shifted to the ward with stable hemodynamics. Bicarbonate correction and hypokalaemia correction were carried out. Oral and maxillofacial surgery (OMFS) opinion was obtained for grade II tooth mobility with mild bleeding, and advice was followed.
Terminal event and outcome
On 18.03.2026, the patient developed hypotension and was started on noradrenaline infusion and shifted to SDICU. Hemoglobin levels dropped again, and one unit of PRBC was transfused. Around 10:15 PM, the patient developed sudden desaturation, requiring re‑initiation of mechanical ventilation. ABG showed metabolic acidosis, and bicarbonate correction was given. Noradrenaline support was escalated. Nephrologist opinion was obtained for decreased urine output, and treatment was initiated. Inj. Aminophylline infusion was started. Poor prognosis was once again explained to the attenders. Subsequently, carotid pulse was not palpable. Cardiopulmonary resuscitation was initiated as per ACLS protocol. Despite all effective measures, return of spontaneous circulation could not be achieved. The patient declared dead on 19.03.2026 at 11:39 PM.
Discussion
Severe hypoglycemia is a well‑recognised neurological emergency and remains a significant cause of morbidity and mortality in patients with diabetes mellitus, particularly in elderly individuals with longstanding disease and associated comorbidities.1 The brain relies almost exclusively on glucose for energy, and prolonged hypoglycemia results in neuronal dysfunction through depletion of intracellular ATP, excitotoxic injury, oxidative stress, and cytotoxic cerebral edema.[7] In the present case, the patient had documented severe hypoglycemia with a random blood glucose of 39 mg/dL, followed by persistent coma and neuroimaging evidence of diffuse cerebral edema. These findings are characteristic of hypoglycemic encephalopathy and are known to confer a poor neurological prognosis, especially when recovery of consciousness does not occur promptly after glucose correction.[6]
This patient’s known hypothyroidism likely contributed to the development, severity, and prolonged course of hypoglycemia. Hypothyroidism is an underrecognized risk factor for hypoglycemia in diabetic patients due to reduced hepatic gluconeogenesis and glycogenolysis, impaired counter‑regulatory hormone responses (cortisol and growth hormone), and decreased insulin clearance, predisposing to recurrent and prolonged hypoglycemia. Furthermore, blunting of autonomic warning symptoms can result in hypoglycemia unawareness and delayed recognition, which may explain the patient’s intermittent slurred speech over the preceding six months, plausibly representing recurrent subclinical neuroglycopenic episodes rather than cerebrovascular events.9
The chronicity of symptoms suggests recurrent hypoglycemic episodes prior to the index event, leading to cumulative neuronal vulnerability, with the subsequent episode of profound hypoglycemia precipitating severe hypoglycemic encephalopathy and secondary hypoxic‑ischemic injury, as evidenced by diffuse cerebral edema on CT and MRI brain imaging. The course was further complicated by systemic inflammatory response, acute kidney injury, hyperammonemia, metabolic acidosis, anemia, and cardiovascular instability, which likely exacerbated hypoglycemia and cerebral injury;[6] despite aggressive intensive care management, including prolonged ventilation, broad‑spectrum antibiotics, vasopressor support, metabolic correction, blood transfusions, and tracheostomy, no meaningful neurological recovery occurred.
Thus, while severe hypoglycemia was the immediate precipitant of encephalopathy in this case, underlying hypothyroidism likely served as an important predisposing factor, contributing to recurrent hypoglycemia, delayed symptom recognition, prolonged neuroglycopenia, and ultimately a fatal outcome. This case highlights the need to consider endocrine comorbidities such as hypothyroidism when evaluating unexplained hypoglycemia and altered sensorium in diabetic patients.
Conclusion
This case illustrates may be a hypothyroidism‑associated hypoglycemic encephalopathy as a devastating but under‑recognised cause of severe neurological injury in patients with diabetes mellitus. The patient’s long‑standing hypothyroidism likely impaired glucose counter‑regulatory mechanisms and insulin clearance, predisposing him to recurrent, possibly unrecognised hypoglycemic episodes, as suggested by a history of intermittent slurred speech over the preceding six months. These antecedent neuroglycopenic events may have culminated in profound hypoglycemia, leading to diffuse cerebral edema, hypoxic–ischemic encephalopathy, and progressive multiorgan dysfunction despite aggressive intensive care management. This case underscores the importance of strict glycaemic surveillance and optimisation of thyroid function in diabetic patients with hypothyroidism, along with early recognition of subtle neurological symptoms, to prevent irreversible neurological sequelae and fatal outcomes.
References
- Zoungas S, Patel A, Chalmers J, de Galan BE, Li Q, Billot L, et al. Severe hypoglycemia and risks of vascular events and death. N Engl J Med 2010;363:1410–8. https://doi.org/10.1056/NEJMoa1003795.
- Kuriyama A, Sato K. Hypoglycemic encephalopathy. CMAJ 2015;187:E437. https://doi.org/10.1503/cmaj.141040.
- Kalra S, Unnikrishnan AG, Sahay R. The hypoglycemic side of hypothyroidism. Indian J Endocrinol Metab 2014;18:1–3. https://doi.org/10.4103/2230-8210.126517.
- Dimitriadis G, Mitrou P, Lambadiari V, Boutati E, Maratou E, Panagiotakos DB, et al. Insulin action in adipose tissue and muscle in hypothyroidism. J Clin Endocrinol Metab 2006;91:4930–7. https://doi.org/10.1210/jc.2006-0478.
- McAulay V, Deary IJ, Frier BM. Symptoms of hypoglycaemia in people with diabetes: Review article. Diabet Med 2001;18:690–705. https://doi.org/10.1046/j.1464-5491.2001.00620.x.
- Barbara G, Mégarbane B, Argaud L, Louis G, Lerolle N, Schneider F, et al. Functional outcome of patients with prolonged hypoglycemic encephalopathy. Ann Intensive Care 2017;7:54. https://doi.org/10.1186/s13613-017-0277-2.
- Auer RN. Progress review: hypoglycemic brain damage. Stroke 1986;17:699–708. https://doi.org/10.1161/01.str.17.4.699.
- Kumar A. Recurrent hypoglycaemia in type 2 diabetic patient due to hypothyroidism. J Diabetes Metab Disord 2015;14:13. https://doi.org/10.1186/s40200-015-0149-y.
- Martín-Timón I, Del Cañizo-Gómez FJ. Mechanisms of hypoglycemia unawareness and implications in diabetic patients. World J Diabetes 2015;6:912–26. https://doi.org/10.4239/wjd.v6.i7.912.