Unmasking hidden hypoxia: A case of methemoglobinemia in a young adult
Sabir Ahamad*, Gowdham Pannirselvam
Department of Nursing, Kauvery Hospital, Marathahalli, Bengaluru
*Correspondence
Abstract
Methemoglobinemia is an uncommon but clinically significant disorder characterized by impaired oxygen delivery due to oxidation of haemoglobin. It often presents with unexplained hypoxia that does not respond adequately to oxygen therapy. This case describes a 25-year-old male who presented with respiratory symptoms and persistent hypoxia. Despite initial management for a suspected pulmonary infection, further evaluation led to the diagnosis of methemoglobinemia. The patient responded well to treatment with methylene blue and supportive therapy. This case emphasizes the importance of early recognition of this condition in patients with disproportionate hypoxia.
Key words: Methemoglobinemia; Hypoxia
Introduction
Methemoglobinemia is a condition in which the iron component of haemoglobin is oxidized from the ferrous to the ferric state, rendering it incapable of binding and transporting oxygen effectively. Under normal physiological conditions, methaemoglobin levels are maintained at less than one percent by endogenous enzymatic reduction systems. However, exposure to oxidizing agents, certain medications, or underlying enzymatic deficiencies can lead to accumulation of methaemoglobin, resulting in tissue hypoxia. Clinically, patients may present with cyanosis and low oxygen saturation that does not improve with supplemental oxygen, making it an important differential diagnosis in cases of unexplained hypoxia.
Case Presentation
A 25-year-old male presented to the emergency department with complaints of cough associated with greenish expectoration for five days, along with fever and cold for two days. He also reported difficulty in breathing, which had progressively worsened. On arrival, the patient was found to be hypoxic with an oxygen saturation of 86% on room air and tachycardic with a pulse rate of 110 beats per minute. Immediate management was initiated with high-flow oxygen at 12 Liters per minute, nebulization, antibiotics, steroids, and antipyretics. Despite oxygen supplementation, the improvement in oxygen saturation was minimal, reaching only 89%, which raised suspicion of an alternative cause of hypoxia.
Past history and clinical examination
The patient had a history of childhood asthma but was otherwise healthy with no significant comorbidities. There was no history of known drug allergies. On clinical examination, the patient was conscious, alert, and oriented. His vital signs revealed tachycardia, elevated blood pressure, and a respiratory rate of 22 breaths per minute, while his temperature was within normal limits. Respiratory system examination revealed bilateral rhonchi, indicating airway involvement. Cardiovascular examination showed tachycardia without any murmurs. The abdominal examination was unremarkable, and neurological assessment was normal.
Investigations
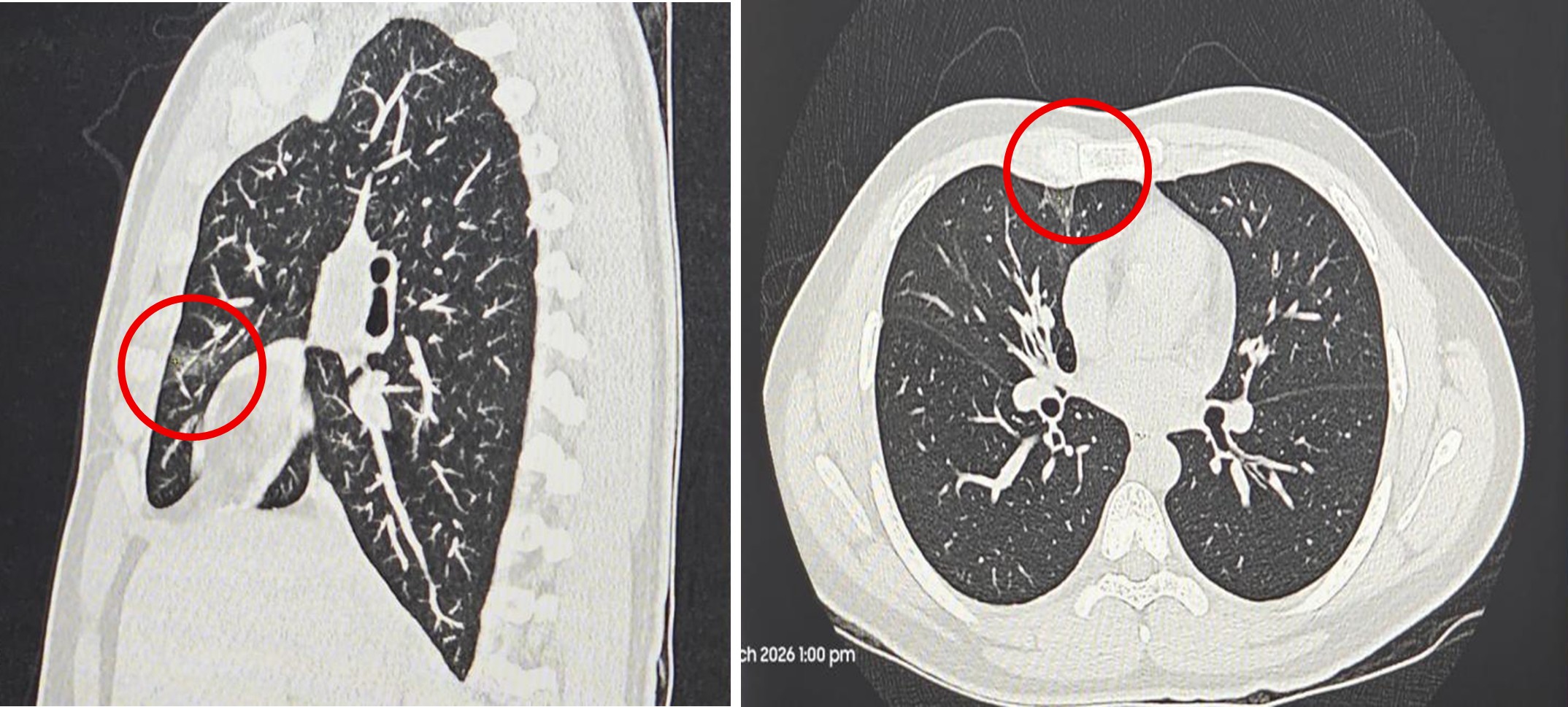
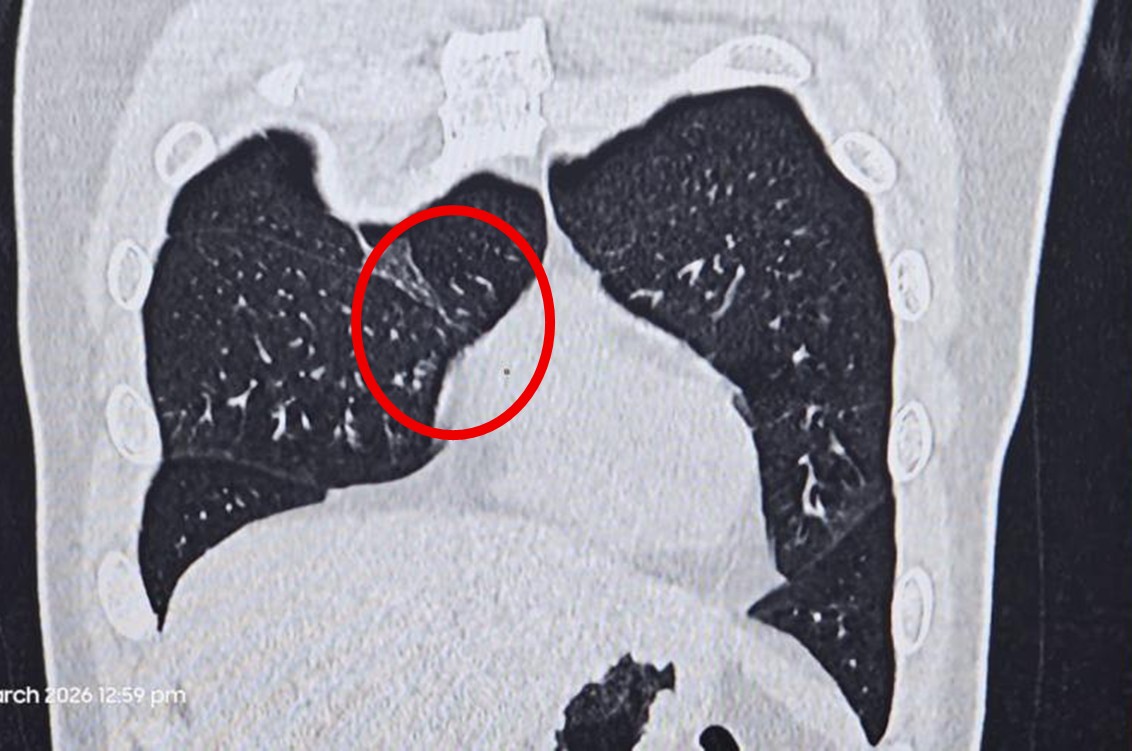
Radiological evaluation with high-resolution computed tomography of the chest revealed small wedge-shaped subpleural ground-glass opacities in the right upper lobe with adjacent thin atelectatic bands, along with subtle ground-glass opacities in the left upper lobe, suggestive of an infective process. Bibasilar discoid atelectasis was also noted.
A chest X-ray demonstrated a normal cardiac silhouette with mild hilar congestion and prominent bronchovascular markings, without any evidence of consolidation or collapse. Laboratory investigations showed an elevated total leukocyte count suggestive of infection, with neutrophilic predominance on peripheral smear, which revealed a normocytic normochromic blood picture. D-dimer levels were mildly elevated, and G6PD levels were within normal limits. Blood cultures were sent for analysis, and viral panel testing was negative.
Diagnosis and pathophysiology
In view of persistent hypoxia that did not respond adequately to oxygen therapy, a diagnosis of Methemoglobinemia was considered. In this condition, oxidation of haemoglobin leads to the formation of methaemoglobin, which is unable to bind oxygen. Additionally, the presence of methaemoglobin causes a leftward shift in the oxygen dissociation curve, impairing oxygen release to tissues. As a result, even though the partial pressure of oxygen in blood may be normal, tissue hypoxia occurs due to reduced oxygen delivery.
Definition
Methemoglobinemia is a clinical condition characterized by an increased level of methaemoglobin in the blood, in which the iron component of haemoglobin is oxidized from the ferrous (Fe²⁺) state to the ferric (Fe³⁺) state. This oxidized form of haemoglobin is incapable of binding and transporting oxygen, leading to impaired oxygen delivery to tissues and resulting in functional hypoxia despite adequate oxygen availability in the blood.
Etiology
Methemoglobinemia can be broadly classified into congenital and acquired forms based on its etiology. Congenital methemoglobinemia arises due to inherited deficiencies in enzymes responsible for reducing methaemoglobin back to haemoglobin, most commonly cytochrome b5 reductase deficiency. In rare cases, abnormal haemoglobin variants such as haemoglobin M may also be responsible.
Acquired methemoglobinemia is far more common and results from exposure to oxidizing agents that overwhelm the body’s natural reduction mechanisms. These agents include certain medications such as local anaesthetics, nitrates, sulfonamides, and antimalarials, as well as environmental and chemical exposures like nitrite-contaminated water, industrial chemicals, and certain toxins. In clinical practice, drug-induced methemoglobinemia is frequently encountered, particularly in hospitalized patients receiving multiple medications.
Pathophysiology
The fundamental abnormality in methemoglobinemia lies in the oxidation of haemoglobin. Under normal conditions, haemoglobin contains iron in the ferrous (Fe²⁺) state, which readily binds oxygen in the lungs and releases it in peripheral tissues. However, when oxidized to the ferric (Fe³⁺) form, haemoglobin becomes methaemoglobin, which cannot bind oxygen.
In addition to this loss of oxygen-carrying capacity, methemoglobinemia also causes a leftward shift in the oxygen-haemoglobin dissociation curve. This means that the remaining normal haemoglobin holds onto oxygen more tightly and does not release it efficiently to the tissues. As a result, there is a dual impairment involving both reduced oxygen transport and decreased oxygen delivery, leading to tissue hypoxia.
The body normally maintains methaemoglobin levels below 1% through enzymatic reduction pathways, primarily the NADH-dependent cytochrome b5 reductase system. A secondary pathway involving NADPH methaemoglobin reductase also contributes, especially under conditions of oxidative stress. When these systems are overwhelmed or deficient, methemoglobin accumulates, leading to clinical manifestations.
Comparison of normal physiology and Methemoglobinemia
| Parameter | Normal Physiology | Methemoglobinemia (Pathophysiology) |
|---|---|---|
| Form of | ||
| Haemoglobin | Haemoglobin contains iron in ferrous (Fe²⁺) state, which can bind oxygen effectively | Haemoglobin is oxidized to ferric (Fe³⁺) state forming methaemoglobin, which cannot bind oxygen |
| Oxygen Binding Capacity | Normal oxygen binding occurs efficiently in the lungs | Oxygen binding is significantly reduced due to dysfunctional haemoglobin |
| Oxygen Delivery to Tissues | Oxygen is released | |
| Appropriately to tissues based on metabolic demand | Oxygen delivery is impaired due to reduced carrying capacity and poor release | |
| Oxygen–Haemoglobin Dissociation Curve | Normal sigmoid curve allowing optimal loading and unloading of oxygen | Leftward shift of the curve leading to increased oxygen affinity and reduced tissue release |
| Methaemoglobin Levels | Maintained below 1% due to efficient enzymatic reduction | Increased levels due to excessive oxidation or defective reduction mechanisms |
| Enzymatic Reduction Mechanism | NADH-dependent cytochrome b5 reductase system maintains balance | Reduction pathways are overwhelmed or deficient, leading to accumulation |
| Response to Oxygen Therapy | Oxygen saturation improves significantly with supplemental oxygen | Poor or minimal improvement despite high-flow oxygen (refractory hypoxia) |
| Blood Oxygen Content | Adequate oxygen content with effective transport and delivery | Functional anaemia due to reduced effective haemoglobin |
| Clinical Appearance | Normal skin coloration and oxygenation | Cyanosis, often with chocolate-brown coloured blood |
| Tissue Oxygenation | Adequate perfusion and oxygen supply to tissues | Tissue hypoxia despite normal arterial oxygen levels |
| Pulse Oximetry Readings | Correlates well with arterial oxygen levels | Shows low saturation with “saturation gap” compared to ABG findings |
| Overall Physiological Outcome | Maintains homeostasis and normal cellular function | Leads to hypoxia, organ dysfunction, and potential life-threatening complications |
Clinical Correlation to the Case
In the presented case, the patient had persistent hypoxia with oxygen saturation of 86% on room air, which improved only minimally despite high-flow oxygen therapy. This poor response to oxygen is a hallmark feature of methemoglobinemia and correlates directly with the underlying pathophysiological mechanisms. The eventual improvement following specific treatment further confirms the diagnosis.
Treatment and mechanism of action
The cornerstone of treatment for methemoglobinemia is the administration of Methylene Blue, which acts as a reducing agent. Methylene blue functions by enhancing the NADPH-dependent pathway of methaemoglobin reduction. After administration, it is converted into leucomethylene blue (LMB) within red blood cells, which donates electrons to methaemoglobin, reducing the ferric (Fe³⁺) iron back to the functional ferrous (Fe²⁺) state. This restores the oxygen-carrying capacity of haemoglobin and improves tissue oxygenation.
The response to methylene blue is typically rapid, with significant improvement in oxygen saturation and clinical symptoms observed within hours. However, its use requires caution in patients with G6PD deficiency, as it may lead to haemolysis due to insufficient NADPH production.
In addition to methylene blue, supportive management plays an important role. This includes administration of high-flow oxygen, although its effectiveness is limited in isolation. Removal of the offending agent is critical in acquired cases. Intravenous fluids, antibiotics, and symptomatic treatment may be required depending on the underlying cause and associated conditions. In severe or refractory cases, advanced interventions such as exchange transfusion or hyperbaric oxygen therapy may be considered.
Management (Case Correlation)
The patient was managed in accordance with the clinical diagnosis of Methemoglobinemia associated with a suspected lower respiratory tract infection. The patient received intravenous Methylene Blue at a dose of 60 mg diluted in normal saline, which is the first-line treatment for methemoglobinemia.
In addition to the specific therapy, the patient was treated empirically for a suspected infectious etiology, as radiological findings revealed ground-glass opacities suggestive of infection. Broad-spectrum intravenous antibiotics, including piperacillin-tazobactam and azithromycin, were administered to cover both typical and atypical respiratory pathogens. Antiviral therapy with oseltamivir was also initiated as part of the management protocol, considering the possibility of viral respiratory infection.
Supportive management played a crucial role in the patient’s recovery. The patient received intravenous corticosteroids in the form of hydrocortisone to reduce airway inflammation, particularly in view of his past history of asthma and the presence of bilateral rhonchi on examination. Nebulization therapy with bronchodilators (Ipratropium bromide and Levosalbutamol) and inhaled corticosteroids (Budesonide) was administered to relieve bronchospasm and improve airway patency. Additional supportive measures included intravenous fluids to maintain hydration, antipyretics for fever control, and proton pump inhibitors for gastric protection.
The patient’s response to treatment was closely monitored through serial oxygen saturation measurements and clinical assessment. Following administration of methylene blue on consecutive days, there was a significant improvement in oxygen saturation from 86% on room air to 94%, and subsequently to 97% at discharge. This clinical improvement strongly correlates with the reversal of methemoglobinemia and effective management of the associated respiratory condition.
Clinical Course
Following treatment, the patient showed gradual improvement in oxygen saturation and overall clinical condition. After receiving methylene blue on two consecutive days, his oxygen saturation improved significantly to 94% on room air. He was subsequently shifted from the intensive care unit to the general ward as his condition stabilized. Over the course of hospitalization, the patient became symptomatically better, was able to tolerate oral intake, and maintained stable hemodynamic parameters.
Condition at Discharge and follow-up
At the time of discharge, the patient was hemodynamically stable with an oxygen saturation of 97% on room air. He was prescribed oral medications including antibiotics, antivirals, corticosteroids, proton pump inhibitors, and vitamin supplementation. Nebulization therapy was continued as part of respiratory management. The patient was advised to maintain a normal diet and gradually resume physical activity as tolerated. Follow-up was scheduled with the pulmonology department after five days, with instructions to seek immediate medical attention in case of recurrence or worsening of symptoms.
Discussion
This case highlights the diagnostic challenge associated with methemoglobinemia, as its presentation can mimic common respiratory conditions. The presence of hypoxia that is disproportionate to clinical and radiological findings and does not respond adequately to oxygen therapy should raise suspicion for this condition. Early diagnosis is crucial, as delayed treatment can lead to severe complications including metabolic acidosis, arrhythmias, and even death. The prompt administration of methylene blue in this case resulted in rapid clinical improvement, underscoring its effectiveness as a therapeutic agent.
Conclusion
Methemoglobinemia is an important but often overlooked cause of refractory hypoxia. Clinicians should maintain a high index of suspicion in patients presenting with unexplained low oxygen saturation, especially when there is poor response to supplemental oxygen. Early recognition and timely treatment can significantly improve outcomes and prevent life-threatening complications.
References
- Chen RJ, Nappe TM. Methemoglobinemia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025.
- Ashurst J, Wasson M. Methemoglobinemia: a systematic review of the pathophysiology, detection, and treatment. Del Med J. 2011;83(7):203–208.
- Cortazzo JA, Lichtman AD. Methemoglobinemia: a review and recommendations for management. J Cardiothorac Vasc Anesth. 2013;27(5):1047–1053.
- McNulty R, Kuchi N, Xu E, Gunja N. Food-induced methemoglobinemia: a systematic review. J Food Sci. 2022;87(4):1423–1448.
- Batton R, Villard S, Popoff B. Methemoglobinemia. Rev Med Interne. 2024;45(8):479–487.
- Ivek I, Knotek T, Ivičić T, Rubinić B, Bajlo P, Hamzić J. Methemoglobinemia: a case report and literature review. Acta Clin Croat. 2022;61(Suppl 1):93–98.
- Wright RO, Lewander WJ, Woolf AD. Methemoglobinemia: etiology, pharmacology, and clinical management. Ann Emerg Med. 1999;34(5):646–656.
- Percy MJ, Lappin TR. Recessive congenital methaemoglobinaemia: cytochrome b5 reductase deficiency. Br J Haematol. 2008;141(3):298–308.
- Umbreit J. Methemoglobin—it’s not just blue: a concise review. Am J Hematol. 2007;82(2):134–144.
- Bura RS, Gorle S, Setti GD, Degala RP. A review of pathophysiology, clinical manifestations, and therapeutic management of methemoglobinemia. J Pharma Insights Res. 2025;3(1):112–120.